
Heat and temperature differ in that heat can beĬonverted to other forms of energy and can be 
If a substance loses heat, again without a change in physical structure, the molecular activity decreases and the temperature drops. If heat is applied to a substance, and there is no change in physical structure (such as lee to water or water to vapor), the molecular activity increases and the temperature rises. Temperature reflects the average molecular activity and is measured by a thermometer on a designated scale, such as the Fahrenheit scale or the Celsius scale. Temperature, although related to heat, is defined as the degree of the hotness or coldness of a substance, determined by the degree of its molecular activity. Heat energy represents the total molecular energy of a substance and is therefore dependent upon both the number of molecules and the degree of molecular activity. Absorption of this energy warms the surface of the earth, and heat is exchanged between the earth's surface and the lower troposphere. Their common source is the radiant energy from the sun. They are never in balance, however, and are constantly undergoing conversion from one form to another, as in the case of the pendulum or the storage battery. When lightning starts forest fires, a similar conversion takes place.Įnergy is present in these various forms in the atmosphere. When the terminals are connected to a resistor, the electrical energy is converted to thermal energy. When a battery is connected to a motor, the electrical energy is converted to mechanical energy in the rotation of the rotor and shaft. When the battery terminals are connected to a suitable conductor, chemical reaction produces electrical energy. The common storage battery in charged condition possesses chemical energy. 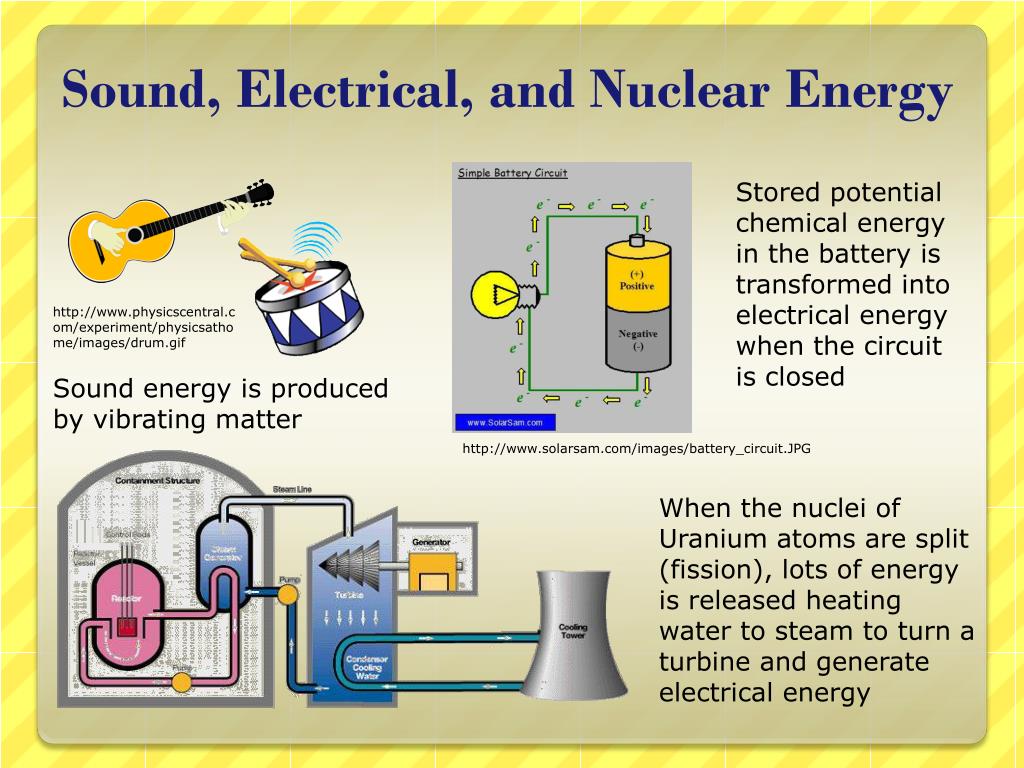
The sun is the earth's source of heat and other forms of energy. Losses caused by friction of the system appear in the form of heat energy. This kinetic energy lifts the pendulum against the force of gravity on the upstroke, and the transformation back to the potential energy occurs. At the end of its swing, a pendulum has potential energy that is expended in the down stroke and converted to kinetic energy. The motion of a pendulum is a good example of the interchange of potential and kinetic energy. Kinetic energy is energy of motion, whereas potential energy is energy due to position, usually with respect to the earth's gravitational field. It cannot be created nor destroyed, although a transformation between energy and mass does occur in atomic reactions. There are also atomic, molecular, and nuclear energy.Įnergy can be, and constantly is being, transformed from one form to another, but energy is always conserved in the process. Its more common forms are heat or thermal energy, radiant energy, mechanical energy (which may he either potential or kinetic), chemical energy, and electrical energy. Simply defined, energy is the capacity to do work. All energy, however, comes either directly or indirectly from the sun. At any time and place, the energy may be in any one form or a combination of several forms. Tremendous quantities of energy are fed into the troposphere, setting it in motion and making it work in many ways to create our everchanging weather.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
